
Explore
When it rains, what’s killing the salmon?
It rains in British Columbia. Water runs off roads, across parking lots, through gutters, and into streams.
Coho salmon are dying. Not from habitat loss. Not from overfishing. Not from climate change — though that isn’t helping. They’re dying from a molecule.
It’s called 6PPD-quinone, and it’s lethal to coho at 0.8 micrograms per litre. That’s parts per billion. A concentration so low you can’t see it, smell it, or taste it. But the salmon can feel it.
Every tyre on every road is shedding this molecule right now.
Tyre rubber contains an antioxidant called 6PPD. It’s there to stop the rubber from cracking — it sacrifices itself to ozone so the tyre doesn’t. Good engineering for the tyre. But when 6PPD reacts with ozone on the road surface, it transforms into 6PPD-quinone.
It sits on the asphalt. It waits. And when it rains, the water carries it into the nearest stream. Every road in the Pacific Northwest is doing this. Every rain event is a pulse of toxin.
No selective binder exists.
Activated carbon — the standard water treatment tool — grabs everything. It doesn’t distinguish between 6PPD-quinone and the thousand other dissolved organics in stormwater. It fills up. It clogs. It can’t be placed in a storm drain and forgotten.
What’s needed is something that grabs this molecule and ignores everything else. Something selective. Something that can sit in infrastructure and work passively, rain after rain.
To design that, you have to look at the molecule itself.
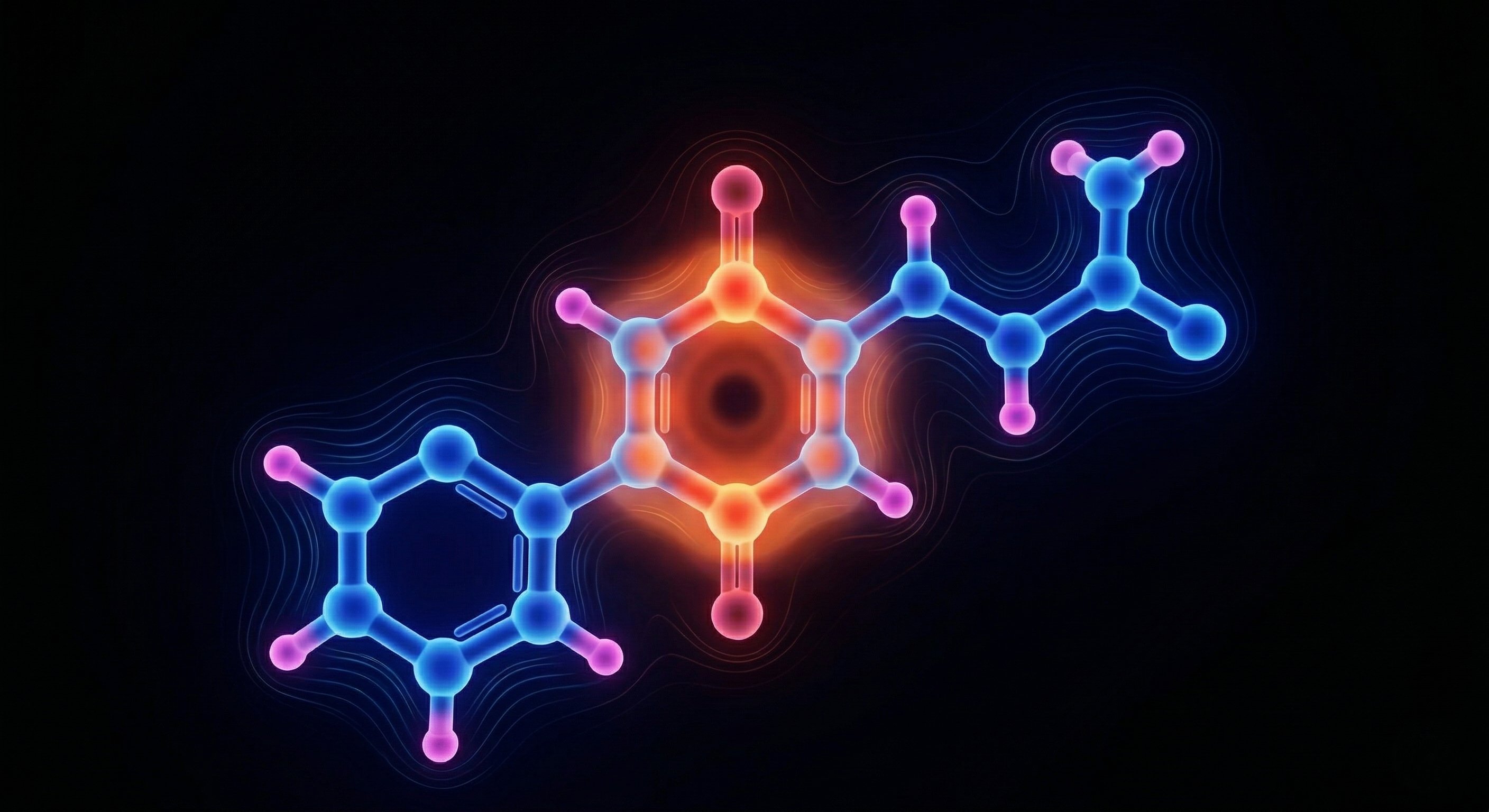
The shape and electronic structure are the design brief.
6PPD-quinone has a quinone ring — electron-poor, hungry for electron density. It has two carbonyl groups positioned para to each other, spaced at a specific geometry. And it has an isobutyl tail — greasy, hydrophobic, wanting to tuck into something non-polar.
Every one of those features is a handle. The electron-poor ring wants electron-rich surfaces. The para spacing sets a geometric constraint. The tail wants a hydrophobic pocket. The molecule is telling you what it needs. The question is whether you can build the thing that provides it.
Interactive
The energy landscape of capture
6PPD-quinone approaching the cage mouth. Watch the energy surface — charge-transfer attraction pulls the quinone ring toward the π-panels, hydrogen bonds lock the geometry, the hydrophobic pocket accepts the tail. A designed minimum on the energy landscape.
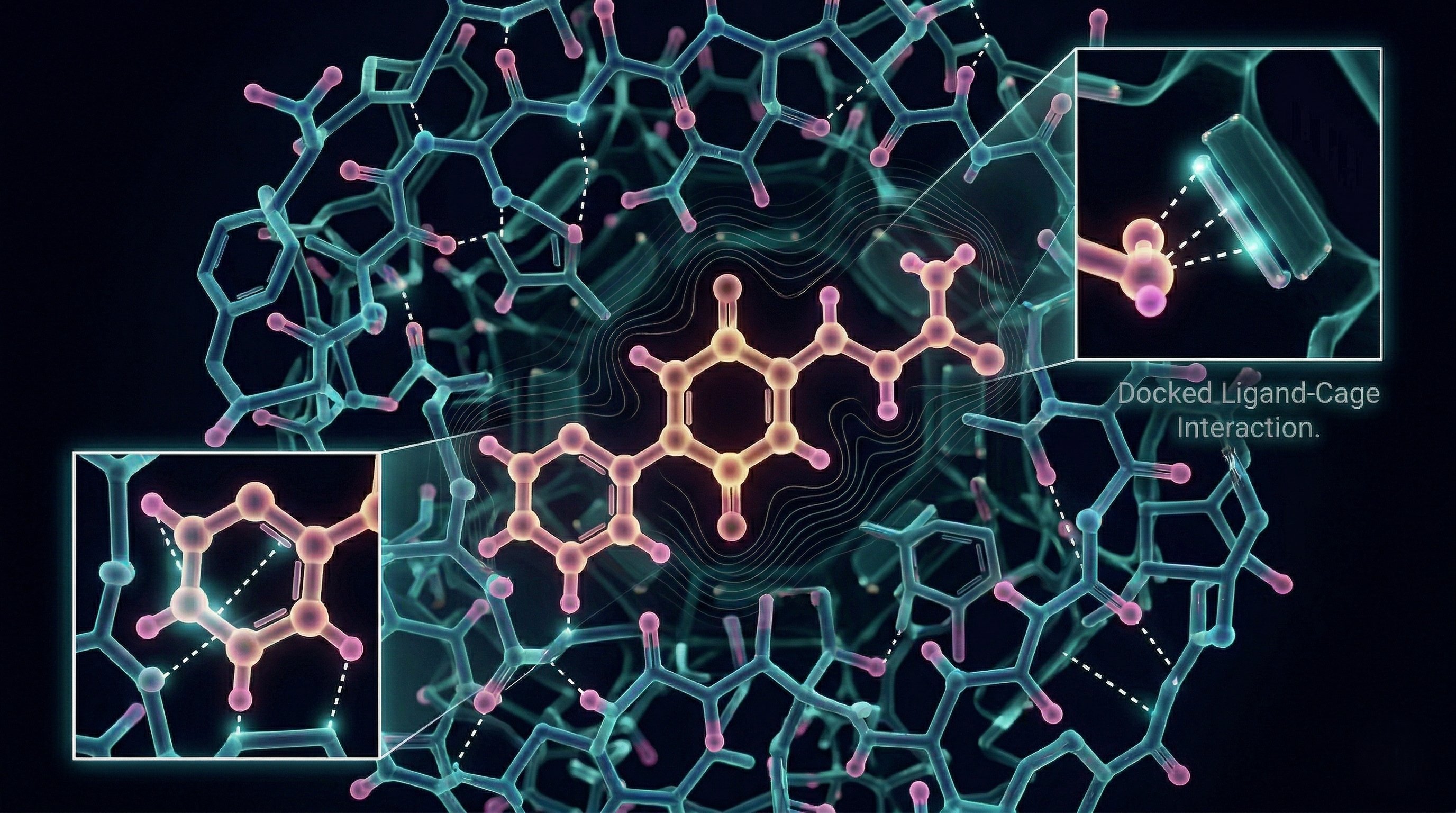
A cage designed from the target’s own physics.
The capture system is a porous organic cage — a molecular container with a designed interior. Electron-rich π-panels line the cavity walls, creating charge-transfer attraction for the electron-poor quinone ring. Urea hydrogen-bond donors sit at 5.4 Å spacing, matching the para-quinone geometry exactly. A hydrophobic pocket accommodates the isobutyl tail.
Every feature of the cage corresponds to a feature of the target. The design didn’t come from screening a library of candidates. It came from reading the molecule’s own energy landscape and building the complementary surface.
What the cage rejects is as important as what it captures.
Stormwater is full of other molecules — PAHs, dissolved organic matter, humic acids. A trap that grabbed everything would be no better than activated carbon. Selectivity doesn’t come from absent attraction. It comes from active repulsion.
The π-panels that attract the electron-poor quinone actively repel electron-rich PAHs. The geometry that matches 6PPD-quinone’s para spacing doesn’t match the symmetry of competing species. The cage isn’t just shaped to accept the target — it’s shaped to reject everything else.
Same principle as the colour stack. The primer rejects unwanted wavelengths. The cage rejects unwanted molecules. Repulsion is half the design.

Click-conjugated to nylon mesh. Placed in a storm drain. Reusable.
The cage is attached to a nylon mesh scaffold using the same orthogonal chemistry that holds the structural dye stack together. The attachment is independent of the cage’s capture function — one chemistry bonds it to the mesh, another captures the toxin. Neither interferes with the other.
The mesh sits in a storm drain. Rain flows through it. 6PPD-quinone is captured. Everything else passes. The scaffold is reusable — the cage doesn’t consume itself in the process. It’s infrastructure, not a consumable. Designed to work passively, rain after rain, season after season.
The same engine that designed a surface to sort photons designed a cage to catch a toxin.
Selective photon capture and selective molecular capture are computationally identical — both reduce to computing how fields interact in confined geometries. One engine handles both. What else can it design a surface to grab?
Why does a drug cost $40,000? →